Innovation
COSMECEUTICALS BASED ON ATEMOYA PEEL EXTRACT: PHOTOPROTECTIVE AND ANTIMICROBIAL ACTION
Cosmecêuticos à base de extrato de casca de atemóia: ação fotoprotetora e antimicrobiana
Tatiane Silva de Abreu1, Pedro Henrique Souza César1, Michelle Carlota Gonçalves2, Roberta Hilsdorf Piccoli2, Silvana Marcussi1
¹ Department of Chemistry, Federal University of Lavras (UFLA) – Lavras, MG, Brazil. 2 Department of Food Sciences, UFLA – Lavras, MG, Brazil.
Abreu, T. S. de
https://orcid.org/0000-0002-3809-5442
César, P. H. S.
https://orcid.org/0000-0003-3271-7797
Gonçalves, M. C.
michelle.goncalves@estudante.ufla.br
https://orcid.org/0000-0002-9567-6905
Piccoli, R. H.
https://orcid.org/0000-0003-2334-9400
Marcussi, S.*
https://orcid.org/0000-0002-4674-6911
*Corresponding author: Biochemistry Laboratory, Department of Chemistry, Federal University of Lavras, Campus Universitário, 37203-202, Lavras, Minas Gerais, Brazil. number: +55 (35) 991509607.
Abstract: Cosmetic formulations, with therapeutic activity, have been the focus of research aimed at determining new applications and adding value to natural products. In this context, plant raw materials stand out for their richness in secondary metabolites, many of which have therapeutic properties. That includes the use of fruits considered unfit for sale, for not meeting appearance standards for fresh fruit purchase and consumption. Thus, the present work developed cosmeceutical emulsions with atemoya peel extract, containing a high content of phenolic compounds and their derivatives with photoprotective, antioxidant and bactericidal action. The emulsions prepared with 2%, 5% and 10% extracts, were within the standards suggested for preliminary stability parameters of cosmetic formulations. All extracts and the emulsion containing 10% atemoya peel, had a sun protection factor equal or greater than 6, and antibacterial activity against Bacillus cereus and Pseudomonas aeruginosa. Finally, they met the ideal physical and sensory standards, presenting good texture and gloss, low stickiness, adequate residual perception, good spreadability and balanced sensation of freshness. The consumer demand for cosmeceuticals with effects for the prevention of premature aging, cancers caused by free radicals and inflammatory diseases, justifies the research and development of multifunctional products, such as the one presented in this study.
Keywords: Annona cherimola Mill., Annona squamosa L., Bioactive cream, Hydroalcoholic extract, Photoprotection.
Resumo: Formulações cosméticas com atividade terapêutica, têm sido foco de pesquisas visando determinar novas aplicações e agregar valor aos produtos naturais. Neste contexto, as matérias-primas vegetais destacam-se pela sua riqueza em metabolitos secundários, muitos dos quais possuem propriedades terapêuticas. Isso inclui a utilização de frutas consideradas impróprias para venda, por não atenderem aos padrões de aparência para compra e consumo de frutas frescas. Assim, no presente trabalho foram desenvolvidas emulsões cosmecêuticas com extrato de casca de atemóia, contendo alto teor de compostos fenólicos e seus derivados com ação fotoprotetora, antioxidante e bactericida. As emulsões preparadas com extratos a 2%, 5% e 10%, estavam dentro dos padrões sugeridos para parâmetros preliminares de estabilidade de formulações cosméticas. Todos os extratos e, a emulsão contendo 10% de casca de atemóia, apresentaram fator de proteção solar igual ou superior a 6, e atividade antibacteriana contra Bacillus cereus e Pseudomonas aeruginosa. Por fim, atenderam aos padrões físicos e sensoriais ideais, apresentando boa textura e brilho, baixa pegajosidade, adequada percepção residual, boa espalhabilidade e sensação equilibrada de frescor. A procura do consumidor por cosmecêuticos com efeitos na prevenção do envelhecimento precoce, dos cânceres causados por radicais livres e das doenças inflamatórias, justifica a pesquisa e o desenvolvimento de produtos multifuncionais, como o apresentado neste estudo.
Palavras-chave: Annona cherimola Mill., Annona squamosa L., Creme bioativo, Extrato hidroalcoólico, Fotoproteção.
INTRODUCTION
Phytocosmetics have shown benefits for the maintenance of human and animal skin health. The use of natural raw materials, combined with concerns regarding skin health care, has motivated the research and development of new cosmetic formulations with reliable safety, efficacy and stability (Surini et al., 2018). These parameters may be associated with the reuse of fruit waste. Therefore, the physicochemical and microbiological stability of an emulsion is a prerequisite for its production. In addition, subtherapeutic activities classify a product as a cosmeceutical.
In this context, atemoya, a hybrid of Annona squamosa L. and Annona cherimola Mill, which belong to the Annonaceae family, is highly produced in Brazil, representing 54% of the Annonaceae market (Brazilian Institute of Geography and Statistics [IBGE], 2017). Among the different parts of atemoya, the fruit peel represents almost 30% of the fruit weight, with higher concentrations of phenolic compounds, potassium and iron than the rest of the fruit; however, the peel is the least studied part (Moraes, 2016). Several secondary metabolites stand out in the composition of the Annonaceae, such as acetogenins, essential oils, alkaloids, terpenoids and flavonoids (Kazman et al., 2022).
Despite the lack of statistical reports of volume loss from the harvest to sale of atemoya fruits (IBGE, 2017), the low homogeneity in the morphological characteristics of the fruits and, the fact that they are highly perishable suggest large losses relative to total production.
In addition to the possibility of using frozen pulp, in the form of yogurts, ice cream or sweets, the peel of fruits that would be rejected for sale may be a target for diversifying the income of small and family farms.
In the context of sunscreens, according to the renowned market analysis company, Exactitude Consultancy, the global market for sun protection products is expected to grow from US$ 15.38 billion in 2023 to US$ 24.70 billion by 2030. Market analyzes show that North Americans are responsible for 40% of the world photoprotector market. Among the 20 strongest companies in the photoprotector market, the following stand out: Johnson & Johnson, L’Oreal Group, Procter & Gamble, Beiersdorf AG, Bayer AG, Unilever, La Roche-Posay, Revlon, Avon Products and Mary Kay (Exactitude Consultancy, 2023).
Like this, we aimed to develop simple formulations containing ethanol extracts obtained from atemoya peel, that are physicochemically and microbiologically stable, with the solvent used and the absence of parabens, contributing to the low toxicity of the formulations. In addition, the antimicrobial and photoprotective properties of the emulsions and their absorption potential, were investigated.
Highlighted, studies that enable the identification of plant compounds with photoprotective action, scientifically supporting the development and commercialization of products that are harmless to the environment, are of great value in the future reduction of environmental impacts, currently attributed to chemical photoprotectors, such as coral bleaching and death of several marine species that inhabit or depend on these habitats for food and/or reproduction (Da Silva et al., 2022).
With this, the standardization of processes useful to an important niche of society and the use of waste, which contributes to the conservation of the environment and its resources and adds value to food production waste, are some of the goals associated with the development of the proposed cosmeceutical.
MATERIALS AND METHODS
Collection of the material and preparation of the extract
A 20 kg lot of atemoya fruits (destined for disposal) were collected in the municipality of Turvolândia, Minas Gerais – Brazil (latitude: 21° 52’ 32” S and longitude: 45° 47’ 13” W) on June 18, 2018. The plant material was identified in the herbarium of the Department of Biology of the Federal University of Lavras (UFLA), where a specimen of atemoya “Thompson” is deposited under record ESAL 30248.
The fruits were cleaned with running water, washed 3 times with 70% ethanol, and pulped, and the peels were dried at 50 °C/60 h until reaching 25% moisture. The dry material was ground in a TE-631 Tecnal mill, and the powder obtained was frozen at -18°C, lyophilized and stored protected from light. The extract was solubilized in 50:50 ethanol: water (v/v).
Alcoholic extraction was performed according to the method described by Moraes (2016). For each 1 g of lyophilized material, 10 mL of solvent (ethanol: water, v/v) was added. Then, the solutions were ultrasonicated for 30 min, macerated overnight and centrifuged at 1500 × g for 15 min. The supernatant was collected, and the precipitate was extracted 2 more times, with 5 mL of solvent, under the same conditions described. The collected supernatants were filtered through Whatman No. 1 filter paper, rotavaped to eliminate ethanol, frozen and then lyophilized.
Formulation of nonionic emulsions
The cosmetic formulation, used for the incorporation of atemoya extracts, was a nonionic emulsion. The latter is compatible with mineral electrolytes and phenolic acids present in the extracts, has low skin irritability and low toxicity and is paraben-free, simple and inexpensive (Table 1).
Table 1
Composition w/w (%) of low cost and paraben-free nonionic emulsions
|
Components (INCI) |
Phase |
Amount p/100 g |
|
Lanette N |
1 |
11 g |
|
Isopropyl myristate |
1 |
3 mL |
|
Polawax |
1 |
3 g |
|
Liquid Vaseline |
1 |
2 mL |
|
Sodium metabisulfite |
2 |
0.3 g |
|
Propylene glycol |
2 |
10 mL |
|
Distilled water |
2 |
Qs 100 mL |
|
Imidazolidinyl urea |
3 |
0.1 g |
|
pH |
6.0 |
INCI: International Nomenclature of Cosmetic Ingredients.
Phases 1 and 2 were separately heated to 75 – 80 °C. Subsequently, phase 2 was poured over phase 1, and the mixture was vigorously stirred, with a mixer for 8 to 10 min, until an emulsion was formed. Imidazolidinyl urea preservative, previously solubilized in water, was added to the emulsion after it reached room temperature by decreasing the stirring speed.
Preliminary stability parameters of the emulsions
Tests to evaluate the stability of the emulsions were applied in the early stages of development of the semisolid formulation, based on the parameters established by the Brazilian Health Regulatory Agency (Anvisa, 2012) and by the Brazilian Pharmacopeia (2019).
Evaluation of organoleptic parameters
The Appearance was evaluated by analysis of phase separation, precipitation and particle agglomeration (samples classified as normal, slightly separated, separated, precipitated or turbid).
The Color was evaluated by comparing a control without extract, with the formulations containing the extract at concentrations of 2%, 5% and 10% (w/w). The color was compared, with a standard, in the same type of flask, against a white light source on a white background.
The Odor was evaluated by comparative analysis between the control and the formulations containing the extract at concentrations of 2%, 5% and 10% (w/w), tested with 30 people. The odor of the sample was compared with the odor of the standard through smell.
Evaluation of physicochemical parameters
The standard samples were kept in a refrigerator (4°C), at room temperature (25°C), or in an oven (40°C), protected from light. The pH was measured using a 10% (w/w) aqueous solution stored for 2 h, and density was determined by the method proposed by the Brazilian Pharmacopoeia (2019).
Mechanical Stress
To assess the resistance to separation of the oil and aqueous phases of the emulsions, 1 g samples were placed in microtubes and subjected to centrifugation at 1000 x g for 30 min at room temperature (Anvisa, 2012).
Heat stress
The samples at concentrations of 2%, 5% and 10% were subjected to heating and cooling cycles. For 12 days, 6 cycles of 24 h at 40 ± 2 °C (in an oven) and 24 h at 4 ± 2 °C (in a refrigerator) were performed, with humidity between 60 and 75% (Anvisa, 2012). At the end of each daily cycle, the organoleptic parameters color, odor, texture, phase separation, consistency, and pH (10% aqueous solution after 2 h of rest) were analyzed.
Microbiological quality control
The microbiological quality of the emulsions containing 2%, 5% and 10% (w/w) atemoya extract was evaluated.
The presence of bacteria was evaluated by dispensing aliquots of the emulsions on soybean tryptone agar (TSA), in Petri dishes, until reaching approximately 3 mm in diameter. The plates were incubated at 37°C for 24-48 h. The presence of fungi and yeasts was assessed using Dichloran Rose-Bengal Chloramphenicol agar. After transferring aliquots of the emulsion samples to the Petri dishes, the dishes were incubated at 25°C for 7 days.
Evaluation of transmembrane permeability in vitro
The evaluation was performed in a Franz cell-like device. The device consisted of a donor compartment, to which an emulsion was added, and a receiver compartment, to which 40 mL of the receiver solution (phosphate buffered saline [PBS]) were added. The 2 compartments were separated by a selective membrane with a 0.45-nm pore size, similar to the porosity of human skin. The duration of migration was 6 h.
The migration of components to the receiver solution was evaluated periodically, across time through spectrophotometry, at a wavelength of 280 nm. The absorbance values obtained for each interval were used to calculate the concentration of the substances that migrated. The calculations were performed using a calibration curve, for which the standard compound had the highest absorption peak of phenolic compounds (Thakker & Chern, 2003). The highest absorption peak at 280 nm was determined based on the parameters of phenolic compounds.
Spectrophotometric sun protection factor of the emulsions
The in vitro SPF (sun protection factor) of emulsions, containing ethanol extract of atemoya peel at concentrations of 2%, 5% and 10% (w/w), was determined by the Mansur method (Mansur et al., 2016). The emulsions were diluted to obtain a final concentration of 0.2 µL mL-1 in ethanol p.a. Subsequently, the absorbance of the solutions was determined using the solvent as the reference.
To determine the wavelength of maximum absorption (λmax) and the maximum absorbance (Absmax), the solutions prepared with the emulsions were scanned between wavelengths of 290 and 400 nm, allowing the measurement of the absorption in the ultraviolet A and B regions (UVA and UVB). Absolute ethyl alcohol was used as a blank. Benzophenone, methyl anthranilate and octyl methoxycinnamate, with absorptions at λmax 288 and 328; 336; and 311 nm, respectively, were prepared in absolute ethanol at a concentration of 2.5 µg mL-1 and used as standards. The absorbance values obtained were applied in the equation (1), determined and described by Mansur et al. (2016), for the conversion to SPF-UVB values.
Equation: SPF = CF. (1)
SPF= Spectrophotometric Protection Factor
CF = Correction Factor = 10 relating to a SPF 4
EE (λ)= Erythemogenic effect of radiation λ in nm according to the Mansur table
I(λ)= Intensity of solar radiation at the λ in nm
Abs(λ)= Spectrophotometric reading of the Absorbance of extracts and controls in the λ in nm.
Antimicrobial activity
The antimicrobial activity of atemoya emulsions was evaluated using the disk-diffusion technique (Clinical & Laboratory Standards Institute [CLSI], 2015) with modifications.
The following microorganisms were used: Bacillus cereus (ATCC 14579), enteropathogenic Escherichia coli (ATCC 35401), Pseudomonas aeruginosa (ATCC 27853), Staphylococcus aureus (ATCC 8593) and Candida albicans (INCQS 40120). The stock cultures, stored in freezing medium (glycerol – 15 mL; bacteriological peptone – 0.5 g; yeast extract – 0.3 g; NaCl – 0.5 g; and distilled water – 100 mL), were activated in tryptone soybean broth (TSB) and incubated at 37°C for 24 h.
After culture, the cultures were standardized at 1.5 × 108 CFU mL-1, using the McFarland scale, and 200 µL aliquots of bacterial and yeast suspensions, previously standardized, were inoculated and spread over TSA. Next, approximately 0.2 g of each emulsion (2%, 5% and 10%) was added to the medium and incubated at 37 °C for 24 h for bacteria and 24 °C for 3 days for yeast. After incubation, the inhibition halos formed were measured using a digital caliper. The controls consisted of a paper disk with 10 µL of chloramphenicol 0.1% (w/v) and ampicillin 0.1% (w/v) for bacteria, and nystatin 0.1% (w/v) for yeast.
Sensory analysis and purchase intention
The sensory analysis of the emulsions was performed using a 1-10 hedonic scale, that evaluates parameters of consumer acceptability of the product and their purchase intention, according to the methodology described by Chorilli et al. (2009). This analysis was performed with 50 tasters between 18 and 35 years of age of both sexes.
The emulsion samples were applied to the back of the hand, and then, the participants completed a standard evaluation form. The analyses were performed after approval of the research protocols by the ethics committee on human research (CAAE 13100719.0.0000.5148).
Statistical analysis
The results are presented as the mean of triplicates ± standard deviation, and means of independent test repetitions when necessary. The data were statistically evaluated by analysis of variance, and the means were compared using the Scott Knott test (p<0.05) using the statistical program R (R Development Core Team, 2011).
RESULTS AND DISCUSSION
Preliminary stability parameters of the emulsion
Ethanol extracts of atemoya peel at concentrations of 2%, 5% and 10% were incorporated into the emulsion. Previous studies have detected 3,4-dihydroxybenzoic acid, vanillic acid, ferulic acid, p-coumaric acid, quercetin, catechin, epicatechin, rutin, chlorogenic acid and K+ in the ethanol extracts of atemoya peel (Moraes, 2016).
There are several studies of phenolic compounds incorporated in cosmeceuticals, such as coffee extracts with antioxidant activity (Xuan et al., 2019). Phenolic compounds, such as p-hydroxybenzoic acid, karmic acid, protocatechuic acid and cinnamic acid, were evaluated for their anti-inflammatory, antimicrobial and antityrosinase activities (Taofiq et al., 2019). The anti-inflammatory and antibacterial potential of hydroxamic acid derivatives has also been documented (Taofiq et al., 2017). This set of biological activities related to the skin makes emulsions, which are rich in phenolic compounds, promising multifunctional cosmeceutical formulations.
This research has shown that in the “antiaging” context, ferulic acid shows photoprotective activity against UVB-induced skin erythema (Saija et al., 2000). Conversely, p-coumaric acid at 30 µg mL-1 inhibited expression in dermal fibroblasts (Seok & Boo, 2015), whereas 500 mM chlorogenic acid after 48 h of exposure to B16 melanoma cells suppressed the levels of melanin (Li et al., 2014). Regarding anti-inflammatory function, 20 µM chlorogenic acid reduced the expression of COX-2 (Chen & Wu, 2014), while topically applied ferulic acid reduced the expression of IL-6 (interleukin-6) (Ambothi et al., 2015). Thus, the study of the bioactive properties of phenolic compounds present in atemoya peel, may contribute to health maintenance and skin repair because they affect cell renewal.
When the phenolic compounds are incorporated into a basic cosmetic cream (1% to 5% extract), daily use allows the absorption and functionalization of high doses; however, there is a need to conduct in vivo and in vitro clinical studies to establish the cosmeceutical potential of the formulations (Taofiq et al., 2017).
The emulsions met the standards required for preliminary stability (Table 2). The intensity of the color and acidity were proportional to the concentration of the extract, and remained stable even under mechanical stress.
Table 2
Evaluation of the organoleptic and physical parameters of the emulsions subjected to preliminary stability tests
|
Parameters |
Emulsion |
|||
|
Control |
Ethanol extract of atemoya peel |
|||
|
2% |
5% |
10% |
||
|
Appearance |
N |
N |
N |
N |
|
Color |
White |
Light beige |
Medium beige |
Dark beige |
|
Odor |
N |
N |
N |
N |
|
pH |
6 ± 0.2 |
5.8 ± 0.2 |
5.8 ± 0.2 |
5.7 ± 0.2 |
|
Density (g.mL-1) |
1.2 |
1.2 |
1.2 |
1.1 |
|
Mechanical stress |
Stable |
Stable |
Stable |
Stable |
N = Normal. The results correspond to the means of triplicates of data obtained for each proportion of extract.
The samples showed changes in color starting at the 5th cooling-heating cycle and changes in consistency starting at the 3rd cycle (Table 3).
Table 3
Evaluation of the organoleptic and physical parameters of the emulsion containing 2% ethanol extract of atemoya peel subjected to heat stress
|
Parameter |
Cooling-heating cycles |
||||||||||||
|
0 |
1 |
2 |
3 |
4 |
5 |
6 |
|||||||
|
Storage (Temperature °C) |
|||||||||||||
|
25 |
4 |
40 |
4 |
40 |
4 |
40 |
4 |
40 |
4 |
40 |
4 |
40 |
|
|
Color |
N |
N |
N |
N |
N |
N |
N |
N |
N |
SM |
SM |
SM |
SM |
|
Odor |
N |
N |
N |
N |
N |
N |
N |
N |
N |
N |
N |
N |
N |
|
Texture |
N |
N |
N |
N |
N |
N |
N |
N |
N |
N |
N |
N |
N |
|
Consistency |
N |
N |
N |
N |
N |
SM |
SM |
SM |
SM |
SM |
SM |
SM |
SM |
|
pH |
5.8 |
5.8 |
5.8 |
5.8 |
5.7 |
5.7 |
5.7 |
5.7 |
5.7 |
5.7 |
5.7 |
5.7 |
5.7 |
N = Normal, SM = Slightly modified. The results correspond to the means of triplicates.
The United States Pharmacopeia (United States Pharmacopeia and National Formulary, 2018) recommends values below 102 CFU/g of formulation for mesophilic microorganisms, molds, and yeasts and an assessment of the presence or not of pathogenic bacteria Pseudomonas aeruginosa, S. aureus and E. coli. The emulsion containing the extract at concentrations of 2%, 5% and 10%, showed microbiological stability for both bacteria and yeasts.
Evaluation of transmembrane permeability in vitro
In dermatological products, the action site is located in the epidermis itself; therefore, the determination of the amount of drug able to reach the circulation has no direct influence on the therapeutic effect except for the purpose of analyzing side effects due to blood absorption. The skin permeation observed in vitro reflects the determinant aspects of the in vivo process and, in most cases, has preliminary application in determining the relative availability of dermatological products in the skin layers.
Phenolic acids from propolis (vanillic, coumaric, caffeic and ferulic acids) and vanillin are able to penetrate the skin epidermis and dermis. The penetration and distribution rate are affected both by the physicochemical characteristics of the active substances and by the physical structure and chemical composition of the semisolid vehicle (Žilius et al., 2013). Studies have also reported the application of a cosmetic serum containing grape seed extract rich in phenolic compounds (Surini et al., 2018).
Fig. 1 shows the simulation of the migration rate of skin compounds from emulsions containing different concentrations of ethanol extract of atemoya peel.
Figure 1
Effect of the concentration of ethanol extract of atemoya peel on transmembrane permeability of emulsions
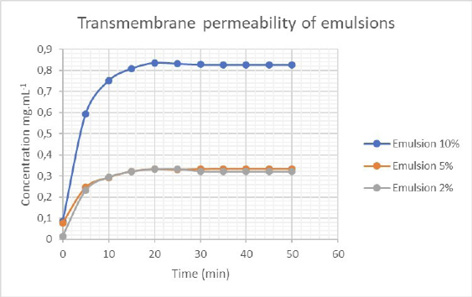
There was no significant difference in transmembrane permeability when comparing the emulsions containing 2% and 5% extract. However, the permeability was significantly higher for the emulsion containing 10% extract, possibly due to the reduction in the physical barrier attributed to the emulsion constituents, and the greater amount of plant metabolites that were able to penetrate the membrane.
Spectrophotometric SPF of the emulsions
A product is considered suitable for use in photoprotective cosmetics when it exhibits an SPF equal to or greater than 6.0 (Anvisa, 2012). All extracts showed satisfactory results; however, when they were incorporated into the emulsions, this minimum parameter was only reached at a concentration of 10% (Table 4).
Table 4
Spectrophotometric SPF of the extract and emulsions containing atemoya peel extract at different concentrations
|
[ ] |
Spectrophotometric SPF |
||||
|---|---|---|---|---|---|
|
Benzophenone |
Methyl anthranilate |
Octyl methoxy cinnamate |
Extract 50:50 (v/v) |
Emulsion |
|
|
5 µg L-1 |
21.1 ± 0.05 |
5.44 ± 0.01 |
29.09 ± 0.02 |
||
|
10 µg mL-1 |
24.35 ± ٠.٠٢ |
7.44 ± ٠.٠٤ |
63.62 ± ٠.٠٢ |
||
|
25 µg mL-1 |
5.56 ± ٠.٠١ |
||||
|
50 µg mL-1 |
8.22 ± ٠.٠٢ |
||||
|
100 µg mL-1 |
13.99 ± ٠.٠٤ |
||||
|
2% |
3.35 ± ٠.٠٣ |
||||
|
5% |
5.32 ± ٠.٠٢ |
||||
|
10% |
8.24 ± ٠.٠٢ |
||||
Controls: Chemical sun filters (benzophenone, methyl anthranilate and octyl methoxycinnamate). Samples: ethanol extract of atemoya peel alone (50:50 v/v, ethanol: water) and emulsions containing different concentrations of the extract. The controls were diluted in absolute ethanol at a concentration of 5 µg mL-1. The emulsions were diluted in 0.2 μg L-1 absolute ethanol and scanned at UVB radiation wavelengths (290 to 320 nm), to calculate the spectrophotometric SPF. The results represent the means of 6 replicates obtained for each concentration and the corresponding standard deviations, * p < 0.05. [ ] = Concentration.
Scientific studies show that phenolic compounds have a common core of benzopyran or chroman bonded to the aromatic rings in the C6-C3-C6 carbon skeleton. Flavonoids dispersed in ethanol show absorption peaks in the UV region, suggesting potential photoprotective activity (Munhoz et al., 2012). These data corroborate the results of the present study.
The major phenolic compounds of atemoya peel are catechins and their derivatives (Moraes, 2016).
Review articles show that EGCG has antioxidant, anti-inflammatory and anticarcinogenic properties and reduces UV-induced erythema/edema. The mechanisms described report the inhibition of lipid peroxidation, the limitation of UV-induced DNA damage, and the reduced production of ROS (reactive oxygen species). In addition, it acts in the suppression of proinflammatory inducers (COX-2 and MMPs) (Souyoul et al., 2018).
Matsui et al. (2009) demonstrated that green tea extract containing polyphenols such as (-)-epigallocatechin-3-gallate (EGCG), despite having a low SPF, showed important secondary characteristics. The action mechanisms occur partly because of their ability to reduce oxidative stress and inhibit NF-kB (nuclear factor kappa B). From this perspective, the emulsion developed, despite having low SPF, has important properties, such as anti-inflammatory, antioxidant and antibacterial properties, desired in a cosmeceutical.
Some studies have shown that green tea polyphenols, applied topically, decrease DNA damage and the number of cyclobutane pyrimidine dimers present in UV-irradiated skin. These dimers are important for initiating UV-induced mutagenesis and carcinogenesis (Elmets et al., 2001; Katiyar et al., 2001; Souyoul et al., 2018).
When graded concentrations of green tea polyphenols, with EGCG as the major polyphenol, were applied to human skin in vivo before exposure to a double minimal erythema dose of solar simulated radiation, there was a dose-dependent reduction in erythema (Elmets et al., 2001). Histologically, a reduction in the hyperproliferation of keratinocytes induced by ultraviolet (UV) light was observed, manifested as a notable decrease in the formation of sunburned cells and a decrease in inflammatory cell infiltrate (Katiyar et al., 2001). In this context, atemoya peel extracts have promising potential for use in the development of cosmetics with photoprotective functions.
The primary factors of photoprotection include physical barriers that reflect and disperse light and chemical barriers that absorb light. Secondary factors include antioxidants, osmolytes (small molecules, such as taurine, that stabilize cells under stressful conditions, regulating hydration) and DNA repair enzymes, which help limit skin damage, disrupting the photochemical cascade that occurs with solar UV light (Campos et al., 2019).
Antimicrobial activity
Table 5 shows the inhibition halos formed by the action of the different emulsions containing atemoya extract on B. cereus and P. aeruginosa.
The 2%, 5% and 10% emulsions did not inhibit the growth of enterogenic E. coli, S. aureus and C. albicans under the evaluated conditions.
Table 5
Antimicrobial activity of cosmetic emulsions containing atemoya peel extract
|
Sample |
Halo diameter (mm) |
||
|
Bacillus cereus |
Pseudomonas aeruginosa |
||
|
*Emulsion |
2% |
4.54 ± 0.68 |
4.53 ± 0.57 |
|
5% |
4.97 ± 1.2 |
5.59 ± 0.62 |
|
|
10% |
6.96 ± 1.05 |
7.39 ± 0.62 |
|
* Supplemented with ethanol extract of atemoya peel (50:50 v/v, ethanol: water) at different concentrations.
The 50:50 ethanol extract of atemoya peel (ethanol: water, v/v) was able to inhibit the growth of S. aureus, S. epidermidis and P. aeruginosa, at concentrations between 75 and 200 mg mL-1 (Abreu, 2020).
Moraes (2016) described in their studies that ethanol extract of atemoya peel had antimicrobial activity against E. coli, B. cereus and S. aureus, and related the activity to the high phenolic content in the peel of this fruit. Moraes (2016) also observed an inhibitory effect of the extract, at the highest concentration evaluated, on S. aureus.
Notably, the ethanol extracts of atemoya peel showed a minimum inhibitory concentration (MIC) of 25 mg mL-1 for S. aureus, S. epidermides and P. aeruginosa; therefore, concentration adjustments should be evaluated to obtain greater effectiveness in the use of the emulsion (Abreu, 2020).
Studies have related the bactericidal action exerted by secondary metabolites to the rupture of the cell membrane of gram-positive microorganisms, and of the cell wall of gram-negative microorganisms. Interactions between flavonoids and peptidoglycans of the bacterial cell wall have been described (Ahmad & Beg, 2001).
Atemoya peel has high concentrations of catechin and epicatechin, which may explain its antimicrobial action, acting as an efficient inhibitor of the enzyme dihydrofolate reductase (DHFR), the target enzyme of antifolate drugs used to treat bacterial infections and, more recently, as an anti-inflammatory agent (Borges et al., 2017). In addition, the high antioxidant activity of these compounds has been described in the literature (Souyoul et al., 2018).
In this context, research on new antimicrobials can significantly contribute to the development of new effective and less toxic products.
Sensory analysis and purchase intention
Based on the data presented (Fig. 2), there was no significant difference between the parameters analyzed.
The 10% emulsion had lower acceptability regarding purchase intention because of its dark beige color. The other characteristics met the ideal physical and sensory standards: good texture and gloss, low stickiness, adequate residual perception, good spreadability and balanced sense of freshness.
Figure 2
Hedonic test of sensory attributes of and purchase intention for emulsions containing 2% (a), 5% (b) and 10% (c) atemoya peel extract, with 50 tasters aged between 18 and 35 years.
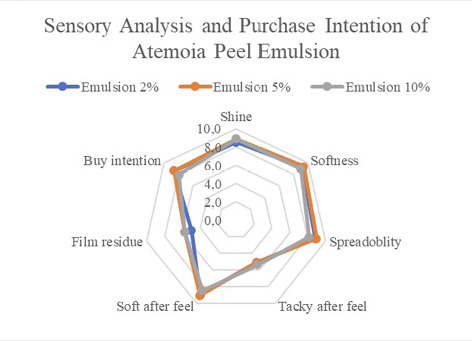
The results represent the means and the corresponding standard deviations, p<0.05.
CONCLUSIONS
In summary, the results found showed the importance of techniques used in this study for the development of a stable formulation with low toxicity, free of parabens, with important properties, absorbable by the epidermis and with good sensory acceptance.
Importantly, the extract used as raw material, originating from discarded fruits, production waste from family agriculture, and its use in the development of cosmetic formulations can add value to fruit waste that would be discarded by providing economic benefits for those in the agricultural production chain, ecological benefits by reducing the environmental impact, and consumer benefits in regard to skin care.
Complementary studies on the properties of atemoya byproducts may contribute to the development of gels, ointments, photoprotectors and other cosmeceuticals.
Acknowledgement and Financial support
Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Nível Superior (Capes), and Fundação de Amparo à Pesquisa do Estado de Minas Gerais (Fapemig).
Conflict of interest
The authors declare that they have no conflicts of interest.
REFERENCES
Abreu, T. S. (2020). Prospecção do potencial cosmecêutico da casca de atemoia. [Tese de Doutorado, Universidade Federal de Lavras].
Ahmad, I., & Beg, A. Z. (2001). Antimicrobial and phytochemical studies on 45 Indian medicinal plants against multi-drug resistant human pathogens. Journal of Ethnopharmacology, 74(2),113-123. https://doi.org/10.1016/S0378-8741(00)00335-4
Ambothi, K., Prasad, N. R., & Balupillai, A. (2015). Ferulic acid inhibits UVB-radiation induced photocarcinogenesis through modulating inflammatory and apoptotic signaling in Swiss albino mice. Food and Chemical Toxicology, 82, 72-78. https://doi.org/10.1016/j.fct.2015.04.031
Agência Nacional de Vigilância Sanitária [Anvisa]. (2012). Guia para Avaliação de Segurança de Produtos Cosméticos. (2ª ed.). Editora Anvisa. http://portal.anvisa.gov.br/documents/106351/107910/Guia+para+Avalia%C3%A7%C3%A3o+de+Seguran%C3%A7a+de+Produtos+Cosm%C3%A9ticos/ab0c660d-3a8c-4698-853a-096501c1dc7c
Agência Nacional de Vigilância Sanitária [Anvisa]. (2012). Regulamento Técnico Mercosul sobre Protetores Solares em Cosméticos e dá outras providências. Resolução n. 30, de 01 de Junho de 2012. http://portal.anvisa.gov.br/wps/wcm/connect/e15afe804c58f17fb8f0f8dc39d59d3e/Resolu%C3%A7%C3%A3o+RDC+N%C2%BA+30,+de+1%C2%BA+de+Junho+de+2012.pdf?MOD=AJPERES
Borges, I. V., Cavalcanti, L. S., Figueirêdo Neto, A., Almeida, J. R. G. S., Rolim, L. A., & Araújo, E. C. C. (2017). Identificação da fração antimicrobiana do extrato da Mimosa tenuiflora. Communicata Scientiae, 8(1), 155-164. https://doi.org/10.14295/CS.v8i1.1493
Brazilian Pharmacopeia. (2019). Anvisa. Brasília. (6ª ed.). p. 92-105, 340-345. https://censos.ibge.gov.br/agro/2017/templates/censo_agro/resultadosagro/agricultura.html?localidade=35&tema=76231
Campos, W. R., Souza, D. C. M., Guimarães, D. G., Santos, V. L. A., Gonsalves, A. A., & Araújo, C. R. M. (2019). Mechanochemical synthesis of symmetric acyclic azines and determination of the uvb solar protection factor in vitro. Química Nova, 42(3), 305-312. https://doi.org/10.21577/0100-4042.20170321
Chen, W., & Wu, L. (2014). Chlorogenic acid suppresses interleukin-1β-induced inflammatory mediators in human chondrocytes. International Journal of Clinical and Experimental Pathology, 7(12), 8797-8801.
Chorilli, M., Udo, M. S., Rodrigues, L. A. P., Cavallini, M. E., & Leonardi, G. R. (2009). Avaliação sensorial de formulações fotoprotetoras contendo filtro solar de amplo espectro. Latin American Journal of Pharmacy, 28(3), 383-392.
CLSI Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically. (2015). Approved Standard – 11th Edition. CLSI document M7-A10 [ISBN 1-56238-836-3]. CLSI, 940 West Valley Road, Suite 1400, Wayne, Pennsylvania 19087-1898.
Da Silva, A. C. P., Santos, B. A. M. C., Castro, H. C., & Rodrigues, C. R. (2022). Ethylhexyl methoxycinnamate and butyl methoxydibenzoylmethane: Toxicological effects on marine biota and human concerns. Journal of Applied Toxicology, 42(1), 73-86. https://doi.org/10.1002/jat.4210
Elmets, C. A., Singh, D., Tubesing, K. A., Matsui, M., Katiyar, S., Katiyar, S., & Hasan Mukhtar, H. (2001). Cutaneous photoprotection from ultraviolet injury by green tea polyphenols. Journal of the American Academy of Dermatology, 44(3), 425-432. https://doi.org/10.1067/mjd.2001.112919
Exactitude Consultancy (2023). Mercado de produtos de proteção solar por tipo de produto (loções, sprays, bastões, lenços umedecidos, outros), canal de distribuição (hipermercados e supermercados, lojas de conveniência, varejo online, farmácias, outros) e região, tendências globais e previsão de 2023 a 2030. https://exactitudeconsultancy.com/pt/reports/34555/sunscreening-products-market/#:~:text=O%20mercado%20global%20de%20produtos,durante%20o%20per%C3%ADodo%20de%20previs%C3%A3o
Instituto Brasileiro de Geografia e Estatística [IBGE]. (2017). Censo Agro. Brasília. (6ª ed.). p. 92-105; 340-345. https://censos.ibge.gov.br/agro/2017/templates/censo_agro/resultadosagro/agricultura.html?localidade=35&tema=76231
Kazman, B. S. M. A., Harnett, J. E., & Hanrahan, J. R. (2022). Traditional Uses, Phytochemistry and Pharmacological Activities of Annonacae. Molecules, 27, 3462. https://doi.org/10.3390/molecules27113462
Katiyar, S. K., Afaq, F., Perez, A., & Mukhtar, H. (2001). Green tea polyphenol (–)-epigallocatechin-3-gallate treatment of human skin inhibits ultraviolet radiation-induced oxidative stress. Carcinogenesis, 22(2), 287-294. https://doi.org/10.1093/carcin/22.2.287
Li, H-R., Habasi, M., Xie, L-Z., & Aisa, H. A. (2014). Effect of chlorogenic acid on melanogenesis of B16 melanoma cells. Molecules, 19(9), 12940-1248. https://doi.org/10.3390/molecules190912940
Mansur, M. C. P. P. R., Leitão, S. G., Cerqueira-Coutinho, C., Vermelho, A. B., Silva, R. S., Presgrave, O. A. F., Leitão, A. A. C., Leitão, G. G., Ricci-Júnior, E., & Santos, E. P. (2016). In vitro and in vivo evaluation of efficacy and safety of photoprotective formulations containing antioxidant extracts. Revista Brasileira de Farmacognosia, 26(2), 251-258. https://doi.org/10.1016/j.bjp.2015.11.006
Matsui, M. S., Hsia, A., Miller, J. D., Hanneman, K., Scull, H., Cooper, K. D., & Baron, E. (2009). Non-sunscreen photoprotection: antioxidants add value to a sunscreen. Journal of Investigative Dermatology Symposium Proceedings, 14(1), 56-59. https://doi.org/10.1038/jidsymp.2009.14
Moraes, I. V. M. (2016). Extração de compostos bioativos da gravioleira (Annona muricata L.) e concentração dos extratos por ultra e nanofiltração. Tese, Unicamp. http://taurus.unicamp.br/bitstream/REPOSIP/321387/1/Moraes_IngridVieiraMachadode_D.pdf
Munhoz, V. M., Lonni, A. A. S. G., Mello, J. C. P., & Lopes, G. C. (2012). Avaliação do fator de proteção solar em fotoprotetores acrescidos com extratos da flora brasileira ricos em substâncias fenólicas. Revista de Ciências Farmacêuticas Básica e Aplicada, 33(2), 225-232.
R Development Core Team. (2011). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Viena. http://www.R-project.org
Saija, A., Tomaino, A., Trombetta, D., Pasquale, A., Uccella, N., Barbuzzi, T., Paolino, D., & Bonina, F. (2000). vitro and in vivo evaluation of caffeic and ferulic acids as topical photoprotective agents. International Journal of pharmaceutics, 199(1), 39-47. https://doi.org/10.1016/S0378-5173(00)00358-6
Seok, J. K., & Boo, Y. C. (2015). p-Coumaric acid attenuates UVB-induced release of stratifin from keratinocytes and indirectly regulates matrix metalloproteinase 1 release from fibroblasts. Korean Journal of Physiology and Pharmacology, 19(3), 241-247. https://doi.org/10.4196/kjpp.2015.19.3.241
Souyoul, S. A., Saussy, K. P., & Lupo, M. P. (2018). Nutraceuticals: a review. Dermatology and Therapy, 8(1), 5-16. https://doi.org/10.1007/s13555-018-0221-x
Surini, S., Mubarak, H., & Ramadon, D. (2018). Cosmetic serum containing grape (Vitis vinifera L.) seed extract phytosome: Formulation and in vitro penetration study. Journal of Young Pharmacists, 10(2), 51. https://doi.org/10.5530/jyp.2018.2s.10
Taofiq, O., González-Paramás, A. M., Barreiro, M. F., & Ferreira, I. C. F. R. (2017). Hydroxycinnamic acids and their derivatives: cosmeceutical significance, challenges and future perspectives, a review. Molecules, 22(2), 281. https://doi.org/10.3390/molecules22020281
Taofiq, O., Heleno, S. A., Calhelha, R. C., Fernandes, I. P., Alves, M. J., Barros, L., González-Paramás, A. M., Ferreira, I. C.F.R., & Barreiro, M. F. (2019). Phenolic acids, cinnamic acid, and ergosterol as cosmeceutical ingredients: Stabilization by microencapsulation to ensure sustained bioactivity. Microchemical Journal, 147, 469-477. https://doi.org/10.1016/j.microc.2019.03.059
Thakker, K. D., & Chern, W. H. (2003). Development and validation of in vitro release tests for semisolid dosage forms-case study. Dissolution Technologies, 10, 10-16. https://doi.org/10.14227/DT100203P10
United States Pharmacopeia and National Formulary (USP 41-NF 36). (2018). Rockville, MD: United States Pharmacopeial Convention. https://www.uspnf.com/
Xuan, S. H., Lee, K. S., Jeong, H. J., Park, Y. M., Ha, J. H., & Park, N. S. (2019). Cosmeceutical activities of ethanol extract and its ethyl acetate fraction from coffee silverskin. Biomaterials Research, 23(2), 2. https://doi.org/10.1186/s40824-018-0151-9
Žilius, M., Ramanauskienė, K., & Briedis, V. (2013). Release of propolis phenolic acids from semisolid formulations and their penetration into the human skin in vitro. Evidence-Based Complementary and Alternative Medicine, 2013, 958717. https://doi.org/10.1155/2013/958717